 The person is sedated, given a local anesthetic, or both. The first step in the process is a trial period of spinal cord stimulation. Once in place, their electrical pulses are meant to block pain signals from reaching the brain.īefore securing FDA approval, the technology was put to the test in its new indication in a study of 270 patients with an average of more than 12 years of pain-constituting what the company has dubbed the largest randomized, controlled trial for SCS in people with nonsurgical back pain.Īmong the first 200 participants in the trial, about 85% of those who were implanted with the Proclaim XR system achieved “significant back pain reduction,” according to Abbott, compared to just 7% of those in the control arm, who were treated with a “conservative medical management” approach comprising physical therapy, medications, massages, acupuncture and more.Īltogether, more than 90% of those who underwent SCS treatment reported significant pain relief or improvements in function, with an average pain reduction of nearly 70%. According to The Journal of Neurosurgery, spinal cord stimulator implantation costs are around 60,000 for Blue Cross Blue Shield clients, while Medicare customers have to pay almost 33,000, with annual fees that are anywhere between 6,000 and 22,000. Spinal cord stimulation for chronic back pain uses electrical pulses to stimulate nerves in the spinal cord, with the goal of interfering with the path of pain signals as they travel to the brain. The expanded approval now has it fighting for share with Nevro, which specializes in spinal cord stimulation, and Medtronic, one of the largest medical device makers by revenue. At Abbott, we understand the heavy burden of chronic pain and how it can take over your life. 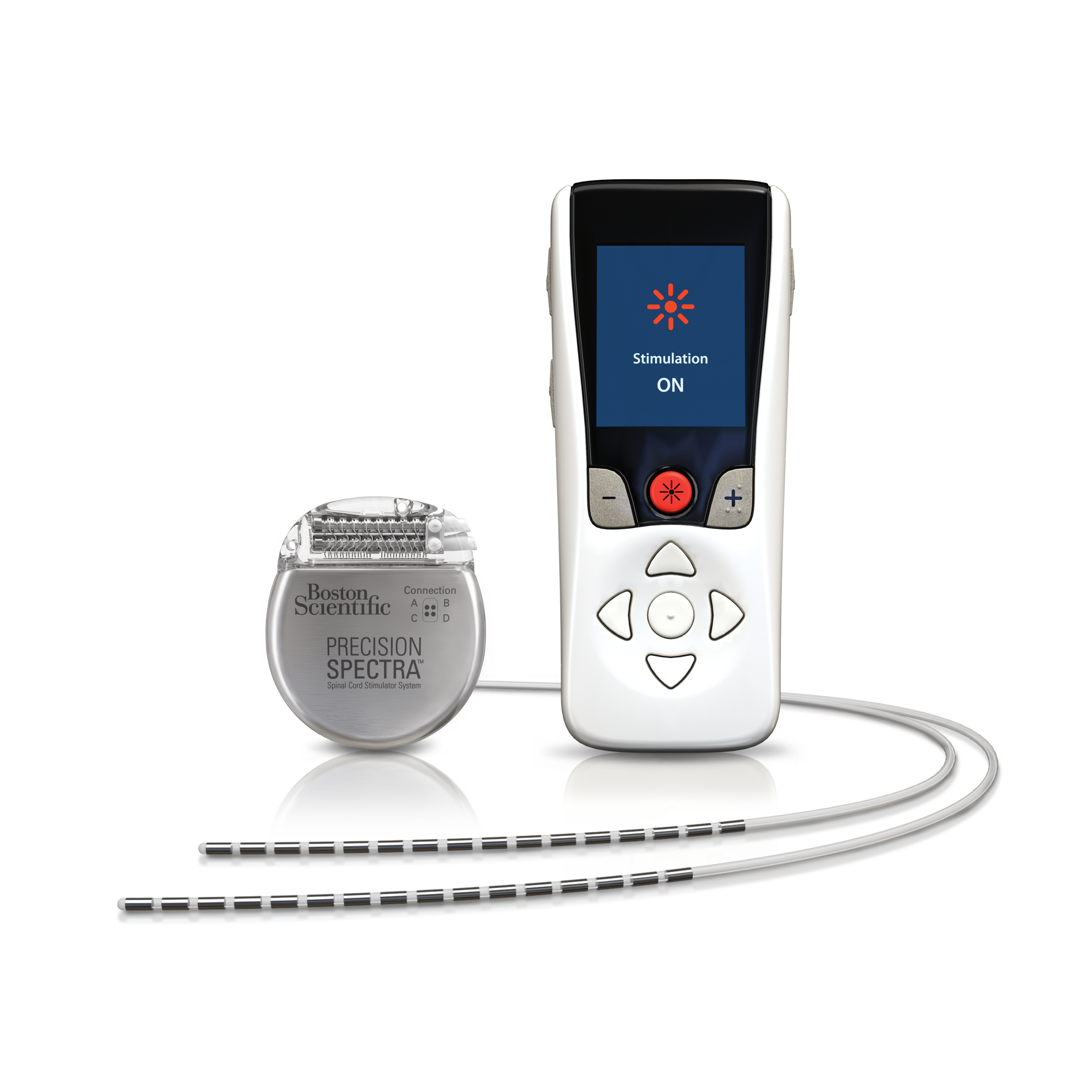
1 Each year about 50,000 people receive SCS therapy, a type of neurostimulation therapy. Abbott has designed the implants so they are safe to use during MRI, provided the devices are put into a special mode that turns stimulation off. Spinal cord stimulation (SCS) therapy has been used for more than 55 years to manage chronic pain and improve quality of life. Dank der neuesten Fortschritte von Abbott haben Ärzte nunmehr Zugang zu zwei weiteren. The affected devices provide stimulation of the spinal cord, dorsal root ganglion and deep brain to treat conditions including chronic pain. In der Vergangenheit hatten Ärzte ausschließlich Zugang zu einer Form der Neurostimulation, der traditionellen tonischen Rückenmarkstimulation (Spinal Cord Stimulation, SCS). 
It sends out short bursts of low-dose electrical energy rather than a continuous stream of stimulation, which Abbott says helps to avoid the uncomfortable tingling feeling that can sometimes occur with the latter “tonic” approach.īurstDR is used in all of Abbott’s SCS devices in the U.S., which center around pulse generators that are implanted under the skin of the lower back, where connected leads can be placed between the spine and vertebrae. Model 3660 / 3662 / 3670 / 3672 (with Penta 5-column paddle lead, 60cm, model 3228) The implantable pulse generator (IPG) for the Proclaim XR SCS System or Proclaim Plus SCS System allows for MR Conditional scans of any body part when the IPG is coupled with MR Conditional leads, implanted in an approved location and a supported RF. Abbott’s Proclaim XR spinal cord stimulation device was first approved to treat chronic pain in 2019. Traditionelle neurostimulation und neurostimulation von abbott im vergleich. Richard Burke Pain & Neuromodulation, PLLC, Best Doctor in Michigan for Spinal Cord Stimulation, Best Doctor in Michigan for Dorsal Root Stimulation, Best Doctor in Northern Michigan for Spinal Cord Stimulation, Best Doc in Michigan for chronic pain, Best Doctor in Northern Michigan for peripheral neuropathy and diabetic foot neuropathy, Four types of diabetic neuropathy, a diabetic neuropathy, a spinal cord stimulator, a stimulator, Abbott burst neurostimulator, abbott burst spinal cord stimulator, Abbott chronic pain, Abbott DRG, abbott drg stimulator surgery, Abbott DRG system, Abbott FDA approval, Abbott laboratories neuromodulation, abbott neuromodulation, abbott neuromodulation location, abbott neuromodulation products, Abbott pain, Abbott pain management, Abbott pain management products, Abbott pain manager, Abbott spinal cord stimulator cost, Abbott spinal cord stimulator surgery, Abbott spinal stimulator, Abbott spine, Abbott spine, Abbott spine clinic, abbott spine inc, Abbott spine products, about diabetic neuropathy.Nevro nabs FDA approval for spinal cord stimulator that uses AI to personalize pain treatmentĪbbott’s BurstDR technology is designed to be a closer mimic of the nervous system’s natural rhythm than other SCS approaches. Fortunately, with certain Abbott neurostimulators, you can safely have an MRI scan when conditions for safe scanning are met. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
